Antigen presenting dendritic cells (DC) represent highly specialized immune cells with a central role in immunity and tolerance induction. DC sense antigens, which are taken-up, processed and presented in the context of MHC molecules to elicit antigen specific T cell responses. Specific DC subsets exist that differ in surface phenotype, function, activation state and anatomical localization, including (i) classical DC type 1 and 2 (cDC1 and cDC2, respectively) in lymphoid and non-lymphoid tissues; (ii) plasmacytoid DC (pDC) in blood that represent the major producers of type 1 interferon and (iii) Langerhans cells (LC), the cutaneous contingent of DC in epidermis.
The helix-loop-helix transcription factor Id2 represents a determining factor for DC development (Hacker et al., 2003; Seré et al., 2012). Id2-/- mice lack LC and cDC1. TGFbeta1-/- mice also lack LC and we show that TGFbeta1 acts upstream of Id2 and induces Id2 expression.
We identified two types of LC: short-term LC and long-term LC cells (Seré et al., 2012). Short-term LC develop from Gr-1+ monocytes under inflammatory conditions and are Id2-independent. Long-term LC arise from bone marrow under steady state and depend on Id2. LC reconstitution after inflammation occurs in two waves: an initial fast wave of Gr-1+ monocyte-derived short-term LC, which is followed by a second wave of bone marrow-derived long-term LC.
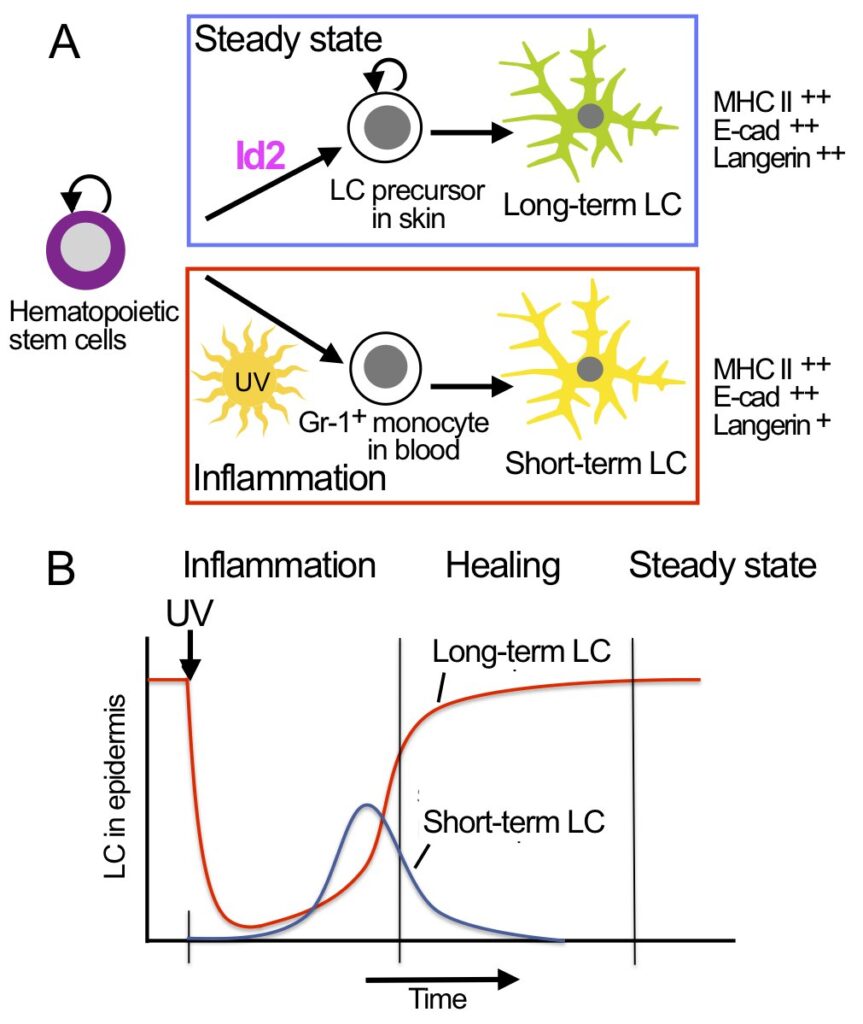
Hematopoietic stem cells and LC precursors in skin develop into long-term LC in steady state, which requires the transcription factor Id2 (A, top panel). In inflammation Gr-1+ monocytes develop into short-term LC, which does not require Id2 (A, lower panel). LC development in inflammation occurs in consecutive waves of short-term LC and long-term LC (B).
Epigenetic and Transcriptional Architecture of Dendritic Cell Development
DC subsets develop from hematopoietic stem cells through consecutive steps of lineage commitment and differentiation: multipotent progenitors (MPP) are committed to DC restricted common DC progenitors (CDP), which differentiate into the specific DC subsets cDC1 cDC2 and pDC. The laboratory studies gene expression and chromatin architecture of the MPP-CDP-cDC/pDC sequel by employing genome wide approaches with RNA-Seq, ChIP-Seq, ATAC-Seq and HiChIP-seq (Hieronymus et al., 2005; Felker et al., 2010; Chauvistré et al., 2014; Lin et al., 2015; Gusmao et al., 2016; Li et al., 2019; in collaboration with Ivan Costa, Institute for Computational Genomics, RWTH Aachen University, Aachen, Germany).
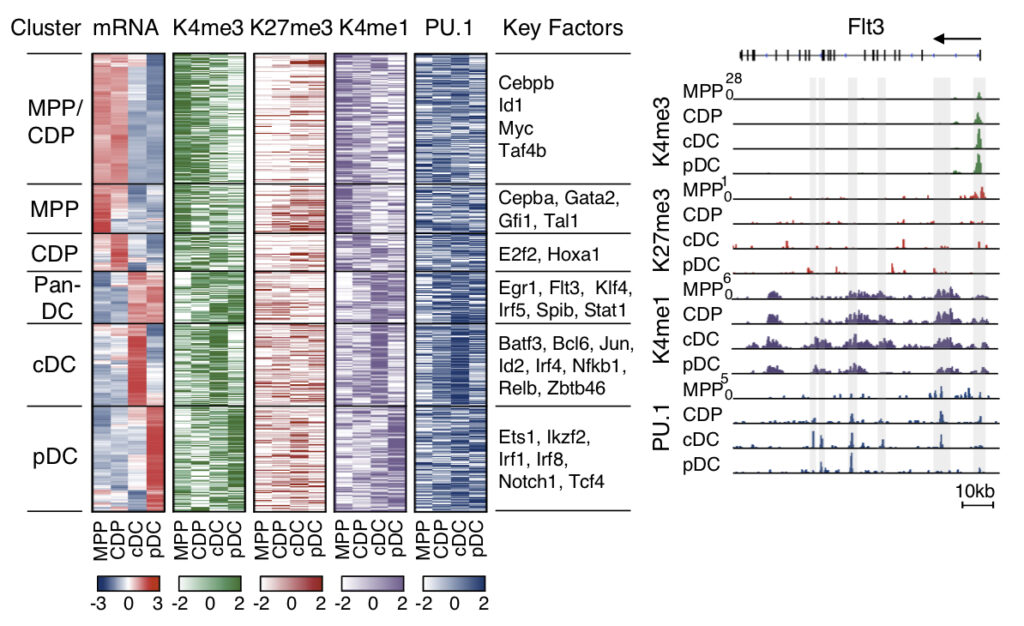
We describe the circuitry of transcription factors, including Irf4, Irf8, Tcf4, Spib and Stats, which drives the sequel MPP-CDP-cDC/pDC. The circuitry also includes positive feedback loops inferred for individual or multiple factors that stabilize the distinct stages of DC development.
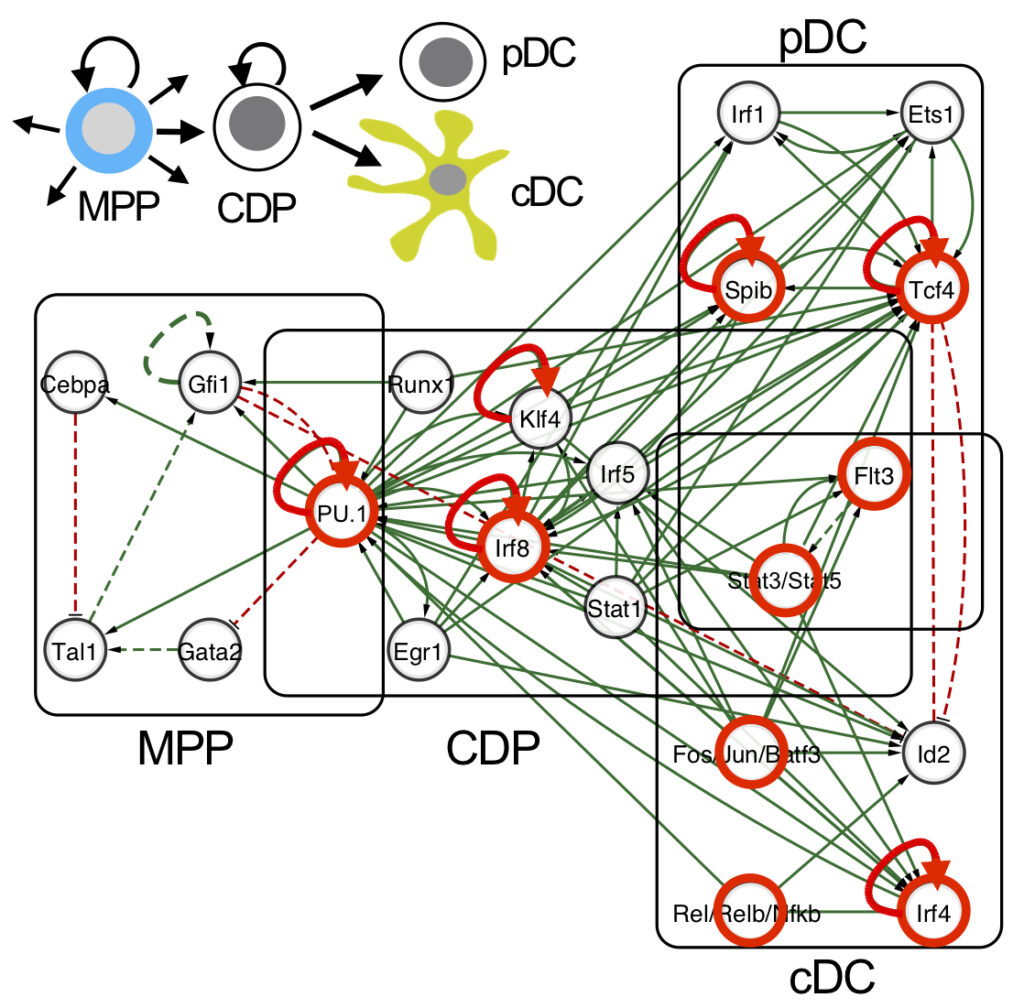
Network illustration of the integrated DC regulatory circuitry of the sequel MPP-CDP-cDC/pDC (Lin et al., 2015). Transcription factors that impact on DC development in gene knockout studies are indicated in red. Feedback loops that stabilize the distinct DC stages in the network are depicted (red arrows).
Data are available by the customized UCSC genome browser track data hub http://www.molcell.rwth-aachen.de/dc/.
Selected Publications
- A lncRNA identifies IRF8 enhancer element in negative feedback control of dendritic cell differentiation.
Xu et al. (2023).
eLife 12, e83342. [Abstract | Full] - Intrathymic dendritic cell precursors promote human T-lineage specification via IRF8-driven transmembrane TNF.
Liang et al. (2022).
Nat. Immunol. 24, 474–486. [Abstract | Full] - Guidelines for mouse and human dendritic cell generation.
Lutz et al. (2023).
Eur. J. Immunol. 53, 2249816. [ Abstract | Full ] - Dendritic cells generated from induced pluripotent stem cells and by direct reprogramming of somatic cells.
Flosdorf and Zenke (2022).
Eur. J. Immunol. 52, 1880–1888. [Abstract | Full] - CRISPR/Cas9 editing in conditionally immortalized HoxB8 cells for studying gene regulation in mouse dendritic cells.
Xu et al. (2021).
Eur. J. mmunol. 52, 1859–1862. [Abstract | Full] - Human DC3 antigen presenting dendritic cells from induced pluripotent stem cells.
Satoh et al. (2021).
Front. Cell Dev. Biol. 9, 667304. [Abstract | Full] - Epigenetic aspects of DC development and differentiation.
Chauvistré and Seré (2020).
Molecular Immunology 128, 116–124. [Abstract | Full] - Identification of transcription factor binding sites using ATAC-seq.
Li et al. (2019).
Genome Biol. 20, 45. [Abstract | Full] - Differentiation of human induced pluripotent stem cells (iPS cells) and embryonic stem cells (ES cells) into dendritic cell (DC) subsets.
Sontag et al. (2017b).
Bio-protocol 7, e2419. [Abstract | Full] - Modelling IRF8 deficient human hematopoiesis and dendritic cell development with engineered induced pluripotent stem cells.
Sontag et al. (2017a).
Stem Cells 35, 898–908. [Abstract | Full] - Epigenetic program and transcription factor circuitry of dendritic cell development.
Lin et al. (2015).
Nucleic Acids Res. 43, 9680–9693. [Abstract | Full] - Two distinct types of Langerhans cells populate the skin during steady state and inflammation.
Seré et al. (2012).
Immunity 37, 905–916. [Abstract | Full] see also Comment by Romani et al. Immunity 37, 766-768 [Abstract | Full] - Transcriptional profiling identifies Id2 function in dendritic cell development.
Hacker et al. (2003).
Nat. Immunol. 4, 380–386, 2003. [Abstract | Full]