In multicellular organisms, specific stem cell types with distinct developmental potentials occur during life. Pluripotent stem cells are generated during early embryonic development and differentiate into derivatives of all three germ layers . Adult stem cells, developing at later stages, are more restricted in their potential and differentiate into cells of only one stem cell system. Adult stem cells occur in all tissues of the adult organism and are important for lifelong tissue homeostasis and repair.
Stem cells are functionally defined by two unique attributes: their high self-renewal capacity and their multilineage differentiation potential. The presence of both characteristics in one cell is rare and sets these highly specialized cells aside from the majority of the somatic cell populations. These unique properties make stem cells ideal targets for stem cell engineering to generate cells with wanted properties.
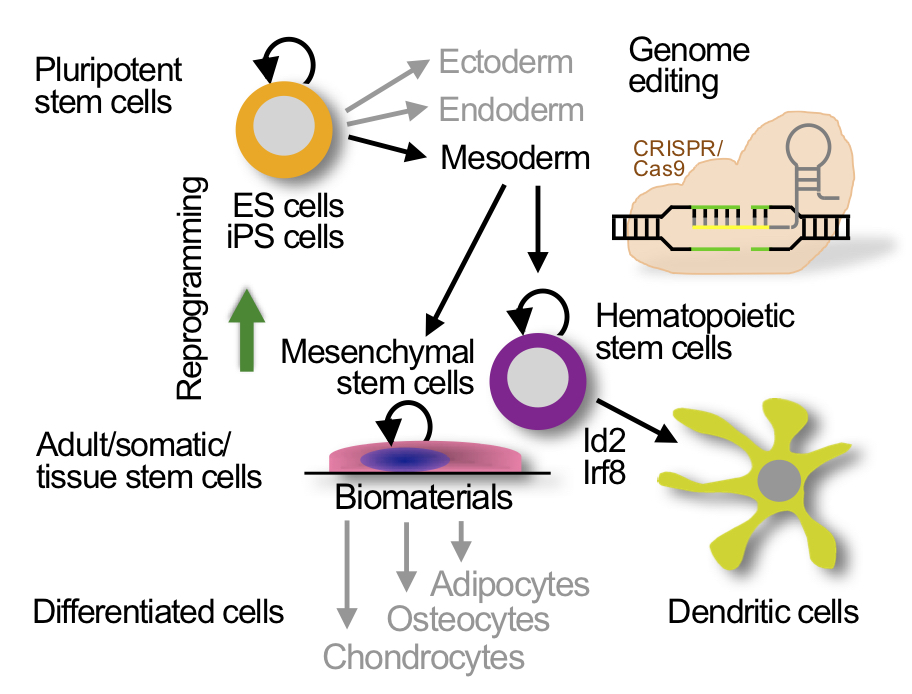
The laboratory studies pluripotent stem cells (ES cells and iPS cells) and adult stem cells (hematopoietic stem cells and mesenchymal stem cells). Patient and disease specific iPS cells are obtained from somatic cells by reprogramming. Hematopoietic stem cells are differentiated into dendritic cells. Genome editing with CRISPR/Cas is used for precision genome engineering.
Gene Expression and Epigenetic Signatures of Hematopoietic Stem Cells
Maintenance of all hematopoietic cells is achieved by the continuous growth and differentiation of multipotent hematopoietic stem cells. We employ transcriptional profiling with RNA-Seq, ChIP-Seq, ATAC-Seq and HiChIP-Seq to determine the gene expression repertoire and chromatin architecture of stem/progenitor cells and their differentiated progeny (Hacker et al., 2003; Hieronymus et al., 2005; Felker et al., 2010; Lin et al., 2015). (see also Epigenetic and Transcriptional Architecture of Dendritic Cell Development).
Stem Cell Engineering
Induced pluripotent stem cells (iPS cells) are obtained by reprogramming of somatic cells with specific sets of transcription factors, such as Oct4, Sox2, Klf4 and c-Myc. We found that Ezh2, a core subunit of Polycomb repressive complex 2 (PRC2), is critical for efficient iPS cell generation (Ding et al., 2014).
iPS cells provide unique opportunities for disease modeling, drug development and cell therapy. However, often their differentiation potential is rather poor. We used cell fusion of iPS cells with hematopoietic stem cells to increase iPS cell differentiation potential towards hematopoietic cells and other mesendodermal lineages, such as cardiomyocytes, hepatocytes and endothelial cells (Qin et al., 2014).
Immunogenicity of iPS cells and iPS cell-derived cells remains controversial. Sertoli cells in testis provide an immune-privileged environment for germ cells. We found that Sertoli cell-derived iPS cells retain some somatic memory of Sertoli cells that confers reduced immunogenicity of iPS cells and iPS cell-derived cells in vivo and in vitro (Wang et al., 2014).
Our data suggest that immune-privileged Sertoli cells represent a preferred source for iPS cell generation if it comes to the use of iPS cell-derived cells for transplantation.
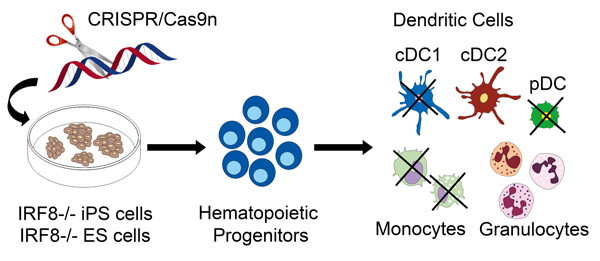
iPS cells and precision genome engineering with CRISPR/Cas are particularly well suited for cell engineering. We used CRISPR/Cas to generated human iPS cells deficient in IRF8 (interferon regulatory factor 8). IRF8 is a lineage determining transcription factor in hematopoiesis and IRF8-/- iPS cell-derived hematopoietic cells are deficient in development of specific DC subsets and in DC function (Sontag et al., 2017a, 2017b).
Patient- and Disease-specific iPS Cells for Drug Discovery
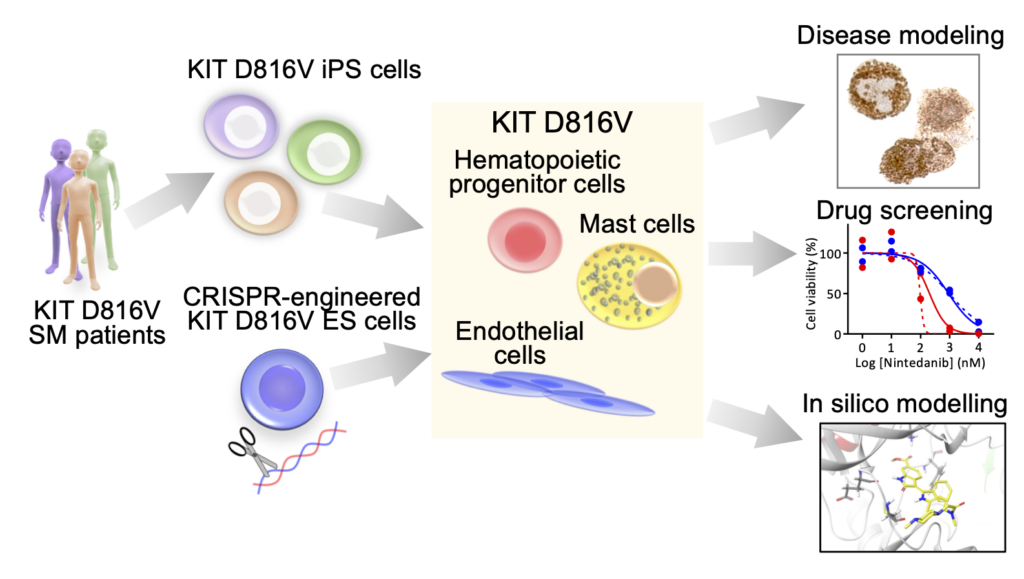
Patient-specific iPS cells allow modelling human disease in vitro (Lampert et al., 2020; Toledo et al., 2021). We generated KIT D816V iPS cells from patients with aggressive systemic mastocytosis and mast cell leukemia to develop disease models for mechanistic and drug discovery studies (Toledo et al., 2021).
Compound screening identified nintedanib, an FDA approved angiokinase inhibitor as a novel KIT D816V inhibitor (Toledo et al., 2021). Our results suggest nintedanib as a new drug candidate for KIT D816V targeted therapy of advanced systemic mastocytosis and mast cell leukemia.
To meet the need of obtaining large numbers of iPS cells for disease modeling and drug development, a consortium of experts in engineering sciences and stem cell biology develops an automatic production system for iPS cells, referred to as StemCellFactory (Elanzew et al., 2020; Ma, Toledo et al. 2022).(www.stemcellfactory.net, www.stemcellfactory.org).
The institute operates a Stem Cell Engineering Unit as a member of the German Stem Cell Cores (German Stem Cell Cores).
For human iPS cells of the institute see https://hpscreg.eu

StemCellFactory III: Dieses Vorhaben wurde aus Mitteln des Europäischen Fonds für regionale Entwicklung (EFRE) gefördert

Selected Publications
- Protocol for generating megakaryocytes from patient induced pluripotent stem cells for disease modelling and compound screening.
Chawla et al., (2026).
STAR-Protocols, in press. - Hijack the HiJAKer: Rethinking therapy of JAK2-mutant MPN.
Zenke and Koschmieder (2025).
Blood 146,2377-2378. [Abstract | Full] - Proinflammatory phenotype of iPS cell-derived JAK2 V617F megakaryocytes induces fibrosis in 3D in vitro bone marrow niche.
Flosdorf et al. (2024). (Cover story)
Stem Cell Reports 19, 224–238. [Abstract | Full] - Automated CRISPR/Cas9-based genome editing of human pluripotent stem cells using the StemCellFactory.
Niessing, Breitkreuz et al. (2024).
Front. Bioeng. Biotechnol. 12, 1459273 [Abstract | Full] - Towards personalized medicine with iPS cell technology: A case report of advanced systemic mastocytosis with associated eosinophilia.
Atakhanov et al. (2022).
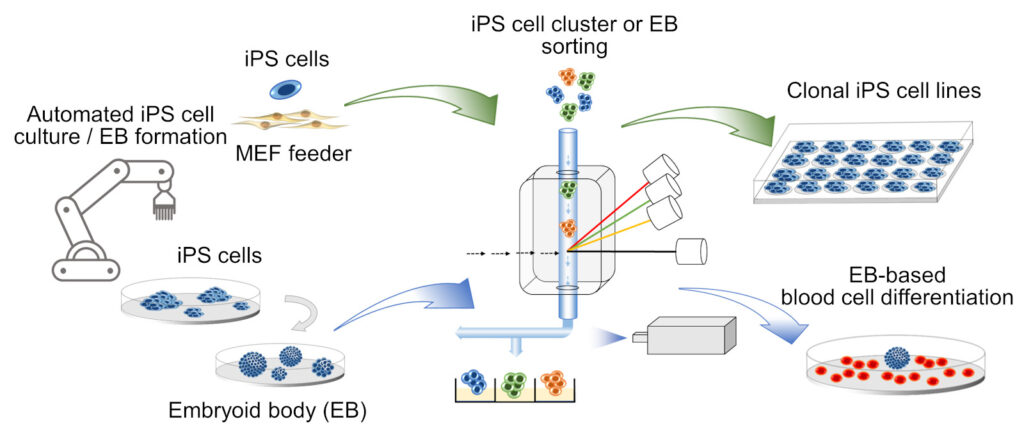
Annals Hematol. 101, 2533–2536. [Abstract | Full] - Cell cluster sorting in automated differentiation of patient-specific induced pluripotent stem cells towards blood cells.
Ma, Toledo et al. (2022).
Front. Bioeng. Biotechnol. 10, 755983. [Abstract | Full] - CRISPR/Cas9 mediated CXCL4 knockout in human iPS cells of polycythemia vera patient with JAK2 V617F mutation.
Boehnke et al.(2021).
Stem Cell Res. 55, 102490. [Abstract | Full] - Nintedanib targets KIT D816V neoplastic cells derived from induced pluripotent stem cells of systemic mastocytosis.
Toledo et al. (2021).
Blood 137, 2070–2084. [Abstract | Full]
(see Commentary by A. Dorrance 2021, Blood 137, 1993–1994) [Abstract | Full] - The StemCellFactory: a modular system integration for automated generation and expansion of human induced pluripotent stem cells.
Elanzew et al. (2020).
Front. Bioeng. Biotechnol. 8, 580352. [Abstract | Full] - The role of Nav1.7 in human nociceptors: insights from human iPS cell-derived sensory neurons of erythromelalgia patients.
Meents, Bressan, Sontag, Foerster et al. (2019).
Pain 160, 1327–1341. [Abstract | Full] - Differentiation of human induced pluripotent stem cells (iPS cells) and embryonic stem cells (ES cells) into dendritic cell (DC) subsets.
Sontag et al. (2017b).
Bio-protocol 7, e2419. [Abstract | Full] - Modelling IRF8 deficient human hematopoiesis and dendritic cell development with engineered induced pluripotent stem cells.
Sontag et al. (2017a).
Stem Cells 35, 898–908. [Abstract | Full] - Reduced immunogenicity of induced pluripotent stem cells derived from Sertoli cells.
Wang et al. (2014).
PLoS ONE 9, e106110. [Abstract | Full] - Cell fusion enhances mesendodermal differentiation of human induced pluripotent stem cells.
Qin et al. (2014).
Stem Cells Dev. 23, 2875–2882. [Abstract | Full] - The polycomb protein Ezh2 impacts on induced pluripotent stem cell generation.
Ding et al. (2014).
Stem Cells Dev. 23, 931–940. [Abstract | Full] - Human adult germline stem cells in question.
Ko et al. (2010).
Nature 465, E1–E3. [Abstract | Full] - Pluripotent stem cells induced from adult neural stem cells by reprogramming with two factors.
Kim et al. (2008).
Nature 454, 646–650. [Abstract | Full] - Pluripotency associated genes are reactivated by chromatin modifying agents in neurosphere cells.
Ruau et al. (2008).
Stem Cells 26, 920–926. [Abstract | Full]